|
6/21/2023 0 Comments Neutral atomProtons are bound together in an atom's nucleus as a result of the strong nuclear force. Protons are a type of subatomic particle with a positive charge. A programmable neutral-atom quantum computer based on a two-dimensional array of qubits led to the creation of 26-qubit GreenbergerHorneZeilinger states and showed the ability to execute. Summary Electrons are a type of subatomic particle with a negative charge. To determine the number of neutrons in this isotope, we subtract 26 from 56: 56 − 26 = 30, so there are 30 neutrons in this atom. In order to be neutral, an atom must have the same number of electrons and protons. The 26 is the atomic number (which is the same for all iron atoms), while the 56 is the mass number of the isotope. Which indicates a particular isotope of iron. Atomic numbers are often listed as a subscript on the left side of an element’s symbol. Given the mass number for a nucleus (and knowing the atomic number of that particular atom), you can determine the number of neutrons by subtracting the atomic number from the mass number.Ī simple way of indicating the mass number of a particular isotope is to list it as a superscript on the left side of an element’s symbol. The mass number of an atom is the sum of the numbers of protons and neutrons in the nucleus. When scientists discuss individual isotopes, they need an efficient way to specify the number of neutrons in any particular nucleus. In fact, there are currently over 3,500 isotopes known for all the elements. Most elements exist as mixtures of isotopes. Dalton thought that all atoms of the same element were exactly the same. The discovery of isotopes required a minor change in Dalton’s atomic theory. Figure 2.6 “Isotopes of Hydrogen” compares the three isotopes of hydrogen. (Round the atomic mass from the periodic table to the nearest whole. 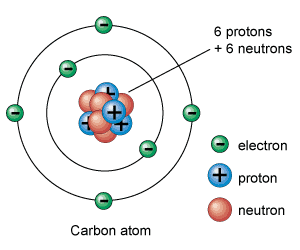
An extremely rare hydrogen isotope, tritium, has 1 proton and 2 neutrons in its nucleus. A neutral atom has an equal number of protons and electrons so that the positive and negative charges exactly balance. Describe which particles and how many of each you need to make a neutral beryllium atom. About 1 in 10,000 hydrogen nuclei, however, also has a neutron this particular isotope is called deuterium. 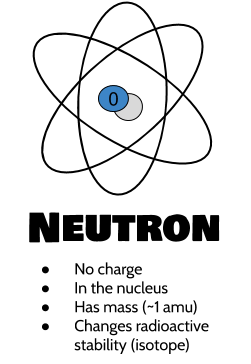
Most hydrogen atoms have a nucleus with only a single proton. Energetic neutral atoms (ENAs) are generated during the charge exchange between energetic trapped, singly charged ions, and ambient cold neutral atoms and. Naturally occurring carbon on Earth, therefore, is actually a mixture of isotopes, albeit a mixture that is 99% carbon with 6 neutrons in each nucleus.Īn important series of isotopes is found with hydrogen atoms. For example, 99% of the carbon atoms on Earth have 6 neutrons and 6 protons in their nuclei about 1% of the carbon atoms have 7 neutrons in their nuclei. Atoms of the same element that have different numbers of neutrons are called isotopes. However, it was found that atoms of the same element can have different numbers of neutrons. How many neutrons are in atoms of a particular element? At first it was thought that the number of neutrons in a nucleus was also characteristic of an element. The largest atoms have over 100 protons in their nuclei. From this table, you can determine the number of protons in the nucleus of any element. 
Chapter 21 “Appendix: Periodic Table of the Elements” lists the elements and their atomic numbers. This is the first sale of a digital quantum computer based on neutral atoms in Europe. There is no such thing as a hydrogen atom with 2 protons in its nucleus a nucleus with 2 protons would be a helium atom. The 100-qubit system will be installed at the DLR Innovation Center in Ulm. Helium has the atomic number 2 all helium atoms have 2 protons in their nuclei. For example, the atomic number for the element carbon (C) is 6, which means that every atom of carbon contains 6 protons. This discovery was so important to our understanding of atoms that the number of protons in the nucleus of an atom is called the atomic number.įor example, hydrogen has the atomic number 1 all hydrogen atoms have 1 proton in their nuclei. Furthermore, different elements have a different number of protons in their nuclei, so the number of protons in the nucleus of an atom is characteristic of a particular element. In other words, all atoms of the same element have the same number of protons. Negative and positive charges of equal magnitude cancel each other out.In the 1910s, experiments with X rays led to this useful conclusion: the magnitude of the positive charge in the nucleus of every atom of a particular element is the same. \)) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly \(1\) in this unit system. We can calculate an atom's formal charge using the equation FC VE - LPE - (BE), where VE the number of valence electrons on the free atom, LPE the number of lone pair electrons on the atom in the molecule, and BE the number of bonding (shared) electrons around the atom in the molecule.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
